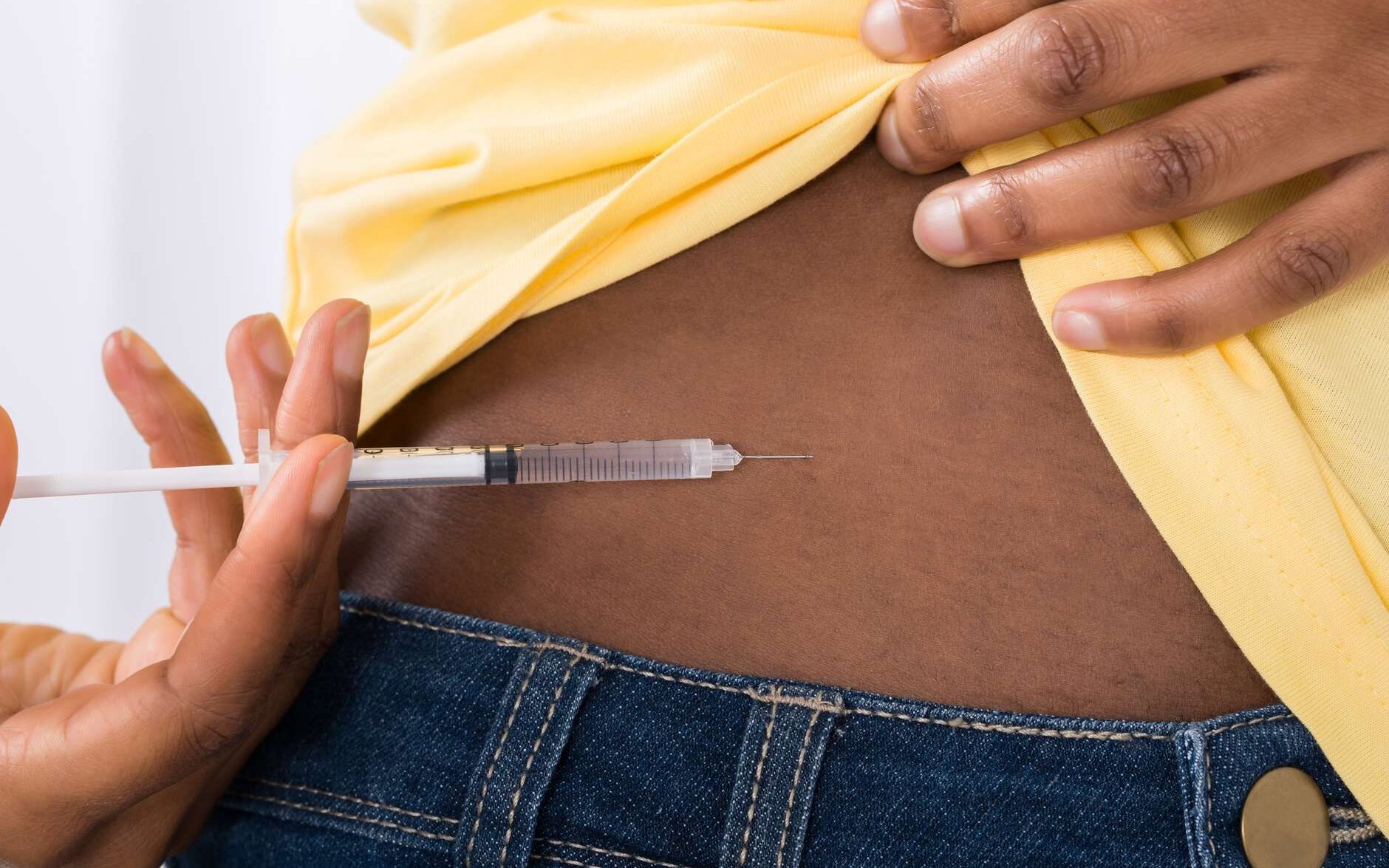
The first Kenyans to receive the new HIV prevention drug Lenacapavir will get it on Thursday when the government officially rolls out the injectable in 178 health facilities across 15 counties.
Lenacapavir, known as LEN, is a long-acting injectable pre-exposure prophylaxis (PrEP) administered once every six months. It will be rolled out alongside cabotegravir (CAB-LA), another long-acting injectable PrEP approved in 2024 after being recommended by the World Health Organisation in 2022.
The two join an oral PrEP pill launched in 2017, giving the country three options for people seeking protection against HIV. PrEP is taken before potential exposure by people not yet infected with the virus. It is neither a vaccine nor a cure.
Ruth Kamau, Programmes Officer at the National Aids and STIs Control Programme (Nascop), said there is high overall readiness for the rollout. The government has identified the facilities, trained healthcare workers to administer the drug and assess eligible clients, and put in place mechanisms to track side effects.
“We understand that no single prevention works for everyone, and different people have different needs; that is why we are giving people a choice when they get to a facility,” she said.
“Just like other PrEP methods, it is best to use condoms alongside LEN to prevent unwanted pregnancy and sexually transmitted infections,” she added.
To be eligible, a client must take a mandatory HIV test at the point of care. Other criteria include weight, those who weigh 35kg or more qualify, screening for recent exposure, medical history and disclosure of current medications.
“Eligibility is a clinical decision that will be determined by the healthcare provider when you go to a facility,” said Kamau.
Clients who meet all criteria are free to choose the type of PrEP they prefer.
Dr Elizabeth Irungu, Jhpiego’s Regional Technical Adviser on PrEP Implementation Science and Service Delivery, told Nation that a negative HIV test is critical to avoid drug resistance among people already living with the virus.
“The people who are already infected with the virus are supposed to be on daily medication. They take about three different drugs. If you put somebody on just one medication, then that increases the likelihood of their virus not being responsive to existing medication. Over time, that could lead to resistance,” she said.
“Our test kits in the country give results within a short time. So, the test will be taken at the point of care,” she added.
Dr Irungu said clients on certain medications, including tuberculosis drugs, sexual performance-enhancing drugs and high cholesterol medications, should be guided by their clinicians on dosage.
“This is about giving Lenacapavir in special situations. It may interfere with how different drugs are broken down in the body. This is why it is important for people going for LEN to let their clinicians know what other conditions they have,” she explained.
“The doctor will know what kind of adjustments to make. They will either give an additional dose of LEN at a separate visit, or reduce or change the doses of the other medications you are taking,” she added.
A presentation at the 13th International Aids Society conference in Kigali, titled Lenacapavir Dosing in Special Situations, noted that TB drugs could lower concentrations of LEN, underscoring the need for clients to disclose their full medical history.
The drug is injected subcutaneously, just below the skin, at the lower abdomen or thighs. On the first day, a client receives two injections and two pills immediately, with two extra pills to take about 24 hours later.
“Taking the injection and the pills gives more protection. If you miss out on the pills, the protection could take longer, up to even a month. You will not need pills for the second dose of injections,” said Dr Irungu.
She stressed the importance of returning for the second dose on time to ensure effectiveness.
Most side effects are localised to the injection site. The drug may form a small lump known as a nodule, which fades as it is absorbed into the body. Other side effects include pain, swelling or redness at the injection site, and nausea.
There were no safety concerns in the two clinical trials. The Purpose 1 study, conducted among women in Uganda and South Africa, showed 100 per cent efficacy in preventing new HIV cases. Purpose 2, conducted in the United States, Argentina, Brazil, Peru, South Africa, Thailand and Mexico, showed 96 per cent efficacy.
Kamau said pregnant and breastfeeding mothers can also receive the drug safely.
“We encourage pregnant and breastfeeding mothers to take LEN. It will actually be one of the ways to prevent mother-to-child transmission of HIV,” she said.
The drug is provided free at the selected health facilities.
hshikanda@ke.nationmedia.com



